Intermittent current interruption (ICI)
Introduction
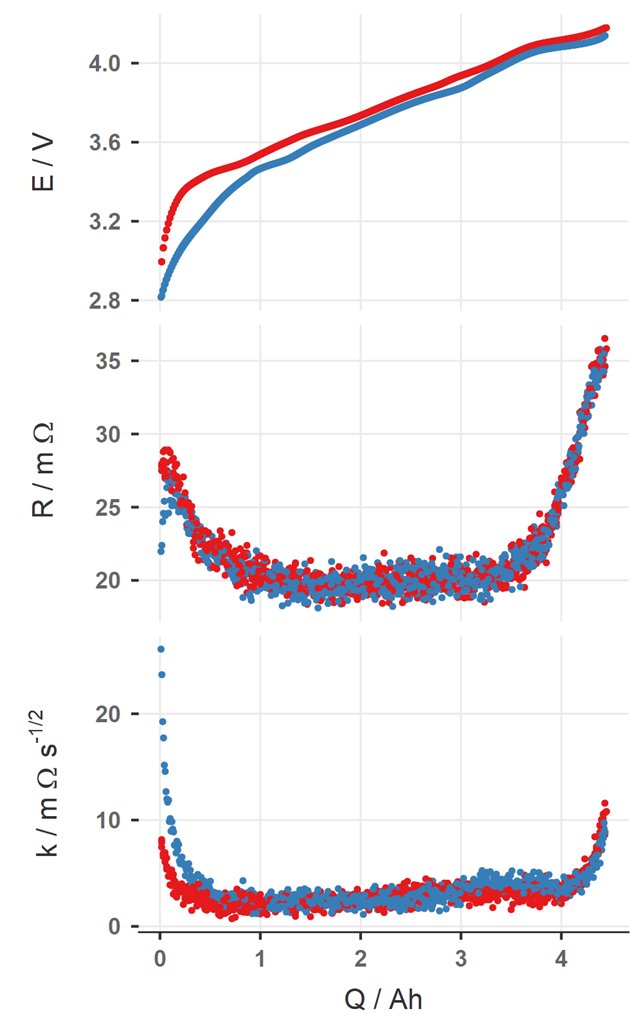
Intermittent current interruption (ICI) is an electrochemical analysis technique for evaluating resistance and energy loss processes in batteries. ICI is a technique I (and my former students at Uppsala University) have driven the development of since 2015, and is intended as a complement to the more well-established direct current internal resistance (DCIR) and electrochemical impedance spectroscopy (EIS) techniques. ICI can also be used as an alternative to the galvanostatic intermittent titration technique (GITT) for diffusion coefficient determination. ICI is, in my experience, ideal for adding an extra dimension to electrochemical characterisation of battery materials as well as an easy, pragmatic method for characterisation of complete battery cells. ICI is particularly well-suited for use in characterising ageing and understanding the variation of resistance as a function of state-of-charge (SoC).
ICI is, simply, the use of regular interruptions in a slow rate (e.g. C/20 OR C/10) constant current charge/discharge cycle, and analysing the potential/voltage change during the interruption to parameterise the resistance in the cell. In this implementation of ICI, in most battery systems the resistance can be parameterised with ICI by two parameters:
- \(R\): the internal resistance, with units of Ω or Ω cm2, is the sum of resistive contributions which obey or can be approximated by Ohm’s law
- \(k\): the diffusion resistance coefficient, with units of Ω s-1/2 or Ω cm2 s-1/2, is a measure of the contribution of mass transport processes to potential drop under load. For a single dominant mass transport process, \(k\) is mathematically related to the diffusion coefficient, which can be determined with careful experimental design.
Pros and cons of ICI
ICI offers the following advantages and disadvantages:
Advantages:
- Fast and convenient. In the majority of use cases, 1-10 second interruptions are sufficient to parameterise the resistance over the entire state-of-charge (SoC) window. A slow charge/discharge cycle incorporating the ICI method adds very little additional experimental time.
- Does not require the use of a frequency response analyser (FRA), or any complex interpretation/software fitting, as would be needed for EIS.
- The parameters \(R\) and \(k\) are, in principle, directly connected to physical phenomena and are independent of current and time, allowing for good transferability between different experiments and systems, which is not the case for DCIR and not always the case for equivalent circuits used to fit EIS data.
- Well-suited for following resistance changes over extended testing and implementing into other routines, such as reference performance tests (RPTs), or other experiments (e.g. in situ/operando characterisation methods).
Disadvantages:
- ICI (in this implementation) requires the response of the system to be diffusion controlled during the measurement time (i.e., E is proportional to t1/2). This covers a vast majority of typical use cases in most systems tested, but not all.
- The simplification of the system necessarily means some processes cannot be directly separated as they can with EIS.
Implementation
ICI can be implemented using any battery cycler which can be programmed to follow a regular loop of constant current steps followed by short interruptions in the current, with a higher data collection rate during the interruption, while still switching between charge and discharge cycles if potential limits are reached. A typical ICI program for a Li-ion battery might look something like this:
- Charge, C/10; step limits: if t > 5 min go to step 2, if E > 4.2 V go to step 3; data point limits dt > 30 s or dE > 5 mV
- Rest 5 seconds, then go to step 1; data point limits dt > 0.2 s
- Discharge, C/10; step limits: if t > 5 min go to step 4, if E < 3.0 V go to step 5; data point limits dt > 30 s or dE > 5 mV
- Rest 5 seconds, then go to step 3; data point limits dt > 0.2 s
- Loop back to step 1, for n cycles
This program would introduce 5 second interruptions every 5 minutes during a constant C/10 charge-discharge cycle, between 3.0 V and 4.2 V.
ICI has been successfully tested on the following brands of battery tester: Bio-Logic, Arbin, Neware, Digatron, Keysight Scienlab, PEC, and likely several more. It has been successfully used to characterise lithium-sulfur batteries, lithium-ion batteries (most major materials, e.g. LCO, NMC, LFP, LNMO, graphite, silicon, conversion materials, lithium metal anodes), sodium-ion batteries and nickel-metal hydride batteries.
The data analysis is performed with a suitable script. I use R, with example scripts in the Supporting Information of some of my papers. I’m aware of other implementations being written in Python, Matlab and Julia.
If anyone else has tried this technique with different potentiostats/programming languages, please let me know!
Background literature
The following four papers from my own publications describe the development of ICI. ICI was initially developed within the scope of lithium-sulfur battery research, and then later transferred over to other chemistries.
[13] M.J. Lacey, K. Edström, D. Brandell, “Visualising the problems with balancing lithium-sulfur batteries by “mapping” internal resistance”, Chem. Commun. 51, 16502-16505 (2015) [view at publisher • post-print • supporting information]
The first paper, which we have since referred to as “ICI 1.0”, is my first report of using an intermittent current interruption to track the resistance changes in a lithium-sulfur (Li-S) battery using either a thick or thin lithium metal foil. At this time, the technique was not referred to as ICI and only considered the voltage drop after a fixed (and arbitrary) time.
[19] M.J. Lacey, “The influence of the electrolyte on the internal resistance of lithium-sulfur batteries studied by an intermittent current interruption method”, ChemElectroChem 4, 8, 1997-2004 (2017) [view at publisher • pre-print • supporting information]
This second paper describes what later became known as “ICI 2.0”, and the first time we used the name “intermittent current interruption”. In this work, each current interruption was analysed by extrapolating the potential E vs t1/2 to zero time after the start of the interruption, calculating the parameter \(R\) from the extrapolated voltage drop at t = 0.
[29] Y.-C. Chien, A.S. Menon, W.R. Brant, D. Brandell, M.J. Lacey, “Simultaneous monitoring of crystalline active materials and resistance evolution in lithium-sulfur batteries”, J. Am. Chem. Soc. 142 (3), 1449-1456 (2020) [pre-print at ChemRxiv, view at publisher, raw data].
In the third paper, the theoretical basis for “ICI 3.0” was developed by Yu-Chuan Chien, calculating the parameter \(k\) from the gradient of E vs t1/2, derived from de Levie’s transmission line model and its connection to the Warburg impedance.
[50] Y.-C. Chien, H. Liu, A.S. Menon, W.R. Brant, D. Brandell, M.J. Lacey, “A fast alternative to the galvanostatic intermittent titration technique”, submitted [pre-print at ChemRxiv].
In the fourth manuscript (currently in peer review), “ICI 4.0” connects the \(k\) parameter obtained for lithium-ion active materials to the solid state diffusion coefficient, from the same theoretical basis as the GITT method.
Other representative examples of use cases
[24] B. Aktekin, M.J. Lacey, T. Nordh, R. Younesi, C. Tengstedt, W. Zipprich, D. Brandell, K. Edström, “Understanding the capacity loss in LNMO-LTO lithium-ion cells at ambient and elevated temperatures”, J. Phys. Chem. C 122 (21), 11234-11248 (2018) [pre-print at ChemRxiv, view at publisher].
Use of ICI (2.0) for following resistance changes of lithium-ion battery materials (LNMO/LTO) in a three-electrode format.
[36] Z. Geng, T. Thiringer, M.J. Lacey, “Intermittent current interruption method for commercial lithium ion batteries aging characterization”, IEEE Transactions on Transportation Electrification 8 (2), 2985 (2021) [pre-print at arXiv, view at publisher].
Validation of ICI (3.0) for large format lithium-ion batteries.
[42] Y.-C. Chien, M.J. Lacey, N.-J. Steinke, D. Brandell, A.R. Rennie, “Correlations between precipitation reactions and electrochemical performance of lithium-sulfur batteries”, Chem 8 (5), 1476-1492 (2022) [pre-print at ChemRxiv, view at publisher].
Combination of ICI (3.0) and operando small angle scattering characterisation to study mass transport limitations in lithium-sulfur batteries.
Other resources
Due to lots of expressions of interest, I have created a tutorial for data analysis using ICI to calculate diffusion coefficients.