Have you read the new perspective article by me, James Frith and Ulderico Ulissi yet?
Just one of the many topics we discuss is the gap between theory and practice in terms of metrics such as energy density, which I’ll briefly elaborate on here.
Specific energy (Wh/kg) and energy density (Wh/L) are two very important metrics for batteries (though far from the only important ones), as they describe the weight or volume required to store a given energy. Batteries are, if we’re honest, not that good at this.
Naturally, when we go looking for new battery materials to increase energy density, we’re looking for combinations with a very high theoretical energy release for their weight or volume. A classic ‘holy grail’ (an overused cliché) is a battery based on lithium and oxygen gas. The reaction 2 Li + O2 <=> Li2O2 (Li-O2 battery) theoretically releases ~3500 Wh/kg (based on the mass of the reactants), which is a lot higher than Li-ion batteries at ~270 Wh/kg today, a comparison you could probably find in many papers on this and similar systems.
Except, this is a completely inappropriate comparison, since we are comparing two different numbers - a materials-level metric and a cell-level figure. The cell-level figure is always (significantly) lower than the materials-level figure, due to several factors.
For cell-level, these factors include the additional materials essential to make the cell function (current collectors, separator, electrolyte, packaging) as well as constraints on the practical reversibility of the chemical reaction.I have heard it said before that as a rule of thumb you can take the theoretical Wh/kg figure and divide by 4 to get the practical maximum. But this rule of thumb doesn’t work, and our article illustrates why this is the case.
In our article, we compare the progression from theoretical (materials-level) to practical (pack-level) for two rather different battery types - small, cylindrical graphite/SiOx | NCA(e.g. Tesla) and large, prismatic, graphite | LFP (e.g. BYD).
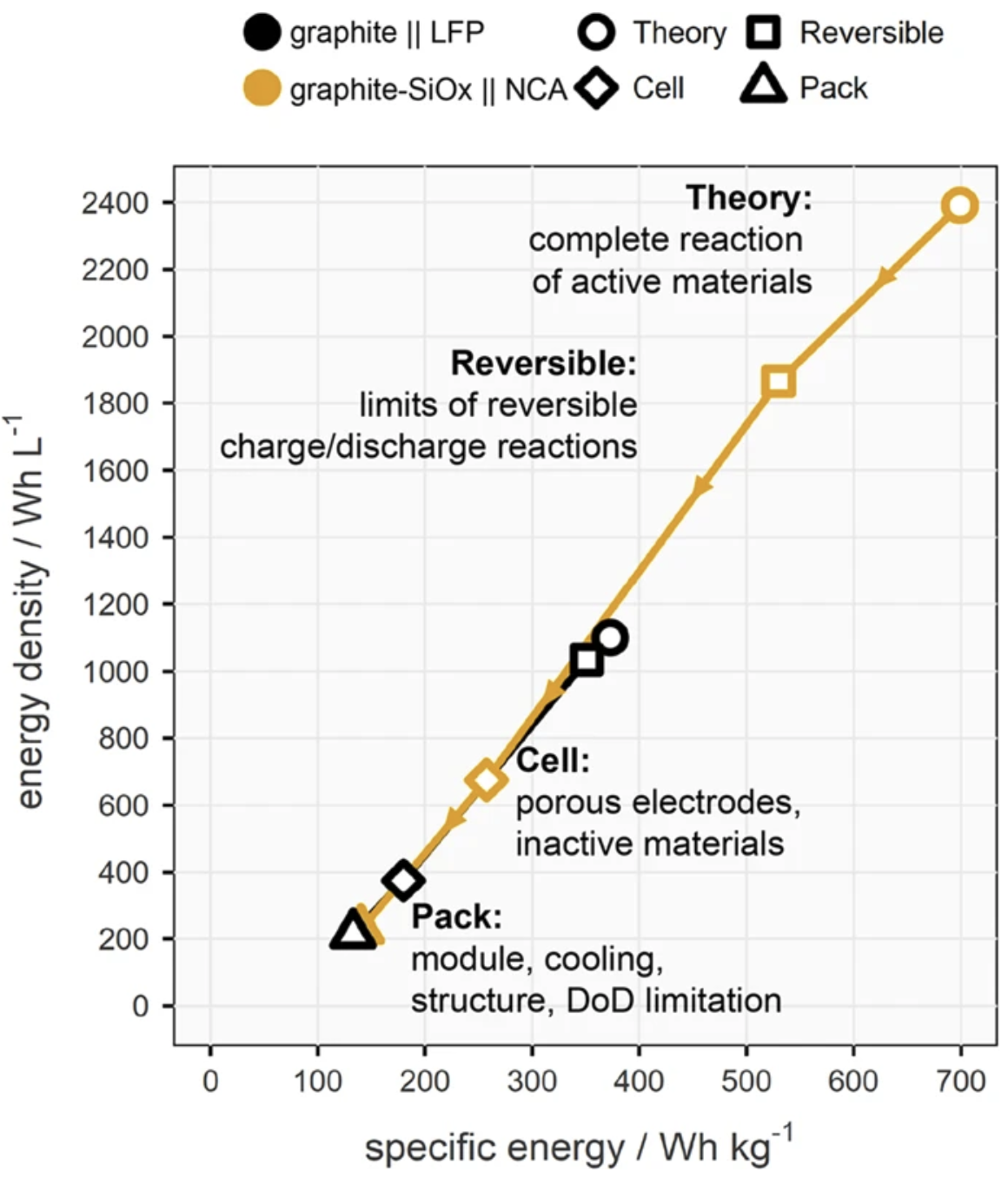
We selected this comparison due to the recent ‘renaissance’ of LFP and the arrival of LFP-based systems with very competitive performance in terms of energy density compared to incumbent NMC/NCA batteries, despite a factor of ~2 difference in theoretical energy density.
There are a number of reasons why the LFP makes up this gap. One is that the reaction can be made reversible closer to the theoretical maximum. But more significant has been the pack-level engineering that takes advantage of LFP’s better thermal stability, which more easily allows for large cells to be sandwiched together in a very volume-efficient way, due to the reduced risk for thermal runaway & propagation.
With this so-called CTP concept we’re now dividing by < 3 for the LFP system from theory to pack level. And since we started writing this article, new products such as CATL’s Qilin pack promise to bring similar benefits to NMC systems and push the envelope further. This also goes to show that there is plenty of mileage in comparatively incremental improvements, especially as we see tighter battery-vehicle integration (e.g. cell-to-car) in the future.
It is also notable that amongst Western automakers, the drive for higher energy density instead led to huge investments in startups developing silicon anodes and solid state batteries, which are not expected to be deployed in production vehicles for several years yet.
For the academic aspect, to briefly return to my Li-O2 discussion from the start of this thread: I think one of the (many) reasons why the research interest in Li-O2 batteries fell sharply in the mid-2010s was the realisation that a Li-air battery, where the system would have to tolerate N2, CO2, H2O and everything else found in air, was extremely unlikely and the cathode would have to be a sealed system with an O2 tank. This would have dragged the energy density down enormously, probably to a level not competitive with technologies at a much higher TRL today.
For future materials discovery, I think it’s important from the start to have a realistic expectation of where practical performance might end up and where the biggest bottlenecks might be found.
Modelling is important here, even a very simple model (e.g. https://github.com/mjlacey/cellmodels/) goes a long way to understanding at least some of the tradeoffs.
What do you think?