The recent controversy over lithium metal calendar life
Felt that my comments on the not-really-a-controversy on lithium metal calendar life, that @stevelevine covered in his recent article, could do with some expanding on. Nuance gets lost in tweets, that’s what threads are for, so here we go…
I had three thoughts on this which I tried to cram into three reply tweets before. First thought was clarifying the 2-3% capacity loss after 24 hours, which is the only thing I could consider to be “controversial” - I think this sentence in the abstract is too strong.
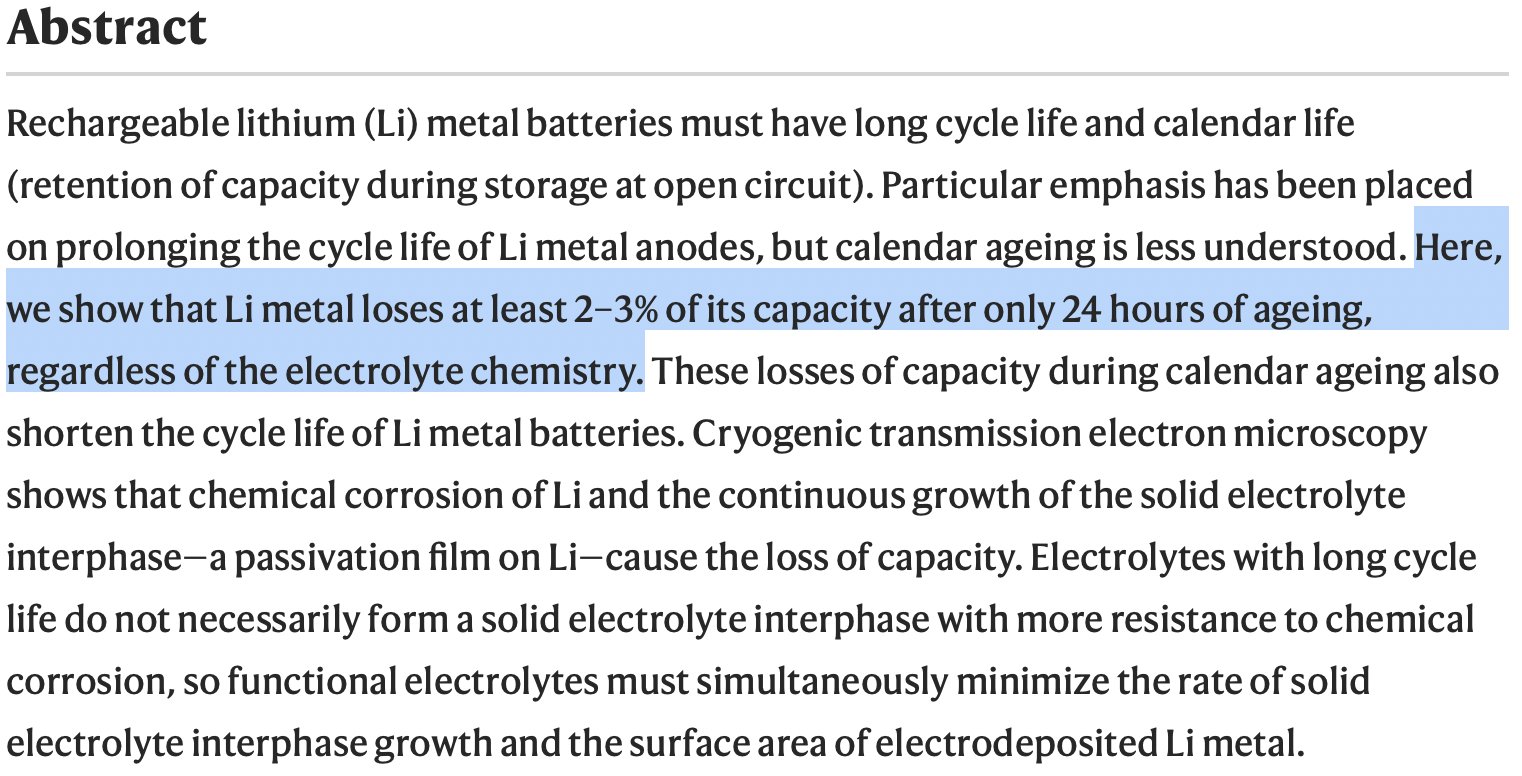
I don’t even think it’s even correct, really, since the results show one electrolyte which loses only 1% capacity; and the result I highlighted in my reply to @stevelevine showed the capacity loss behaviour varies quite a bit across different electrolytes.
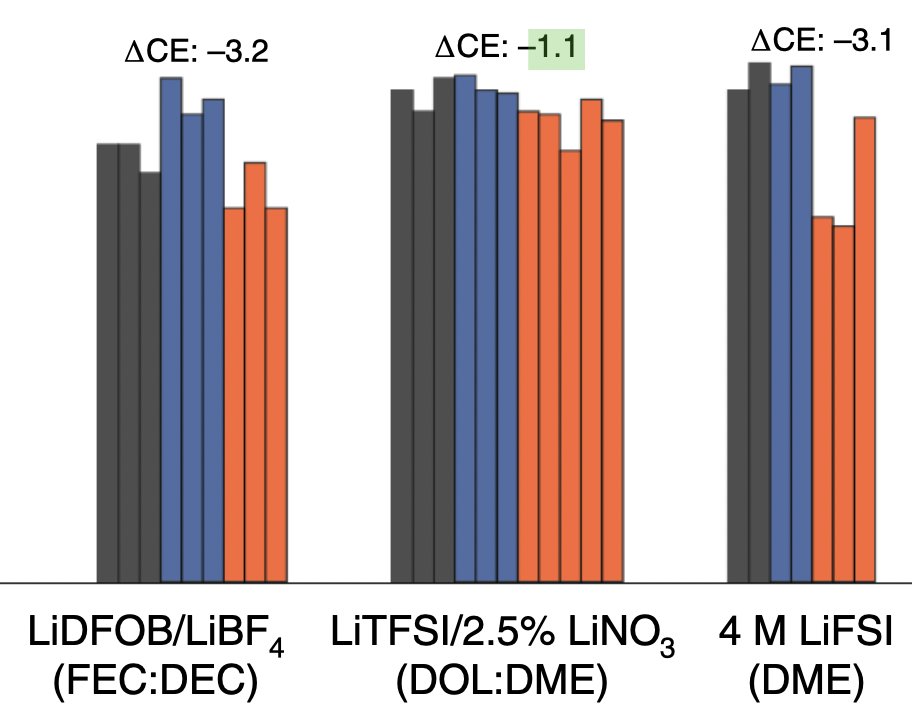
The interesting point in this paper, I thought, was that this capacity loss behaviour (which I rather think of as self-discharge of the lithium electrode) is not clearly correlated with the average efficiency during cycling.
This means that other metrics that one might be tempted to see as indirectly correlated to self-discharge or calendar life, such as cycle life, might not be. Which brings me to my second thought, on lithium-sulfur…
Yes, it’s a different system, but bear with me here. Around 2014 I was looking both at how the Li metal surface changed during self-discharge, and trying to optimise my positive (sulfur) electrode towards something more practically realistic.
I remember reading a lot of papers at that time, mainly on electrode host materials, in which the authors would write about how a major problem of Li-S is that polysulfides diffuse to the anode, which impacts negatively on cycle life.
They would then mainly test the materials by extended battery cycling, and stable cycle life would be given as evidence of good control of polysulfides.
I thought such assertions were sloppy, and a sign of bad control experiments - my baseline electrode at the time, with a decent commercial carbon black and nothing special, could cycle without capacity loss for quite a while, but had terrible efficiency and self-discharge.
So I went to the ISE conference in Taipei in 2015, and early in my talk said that just because you get good cycle life doesn’t mean you don’t have other problems. The point was about appropriate experiments, but it’s also about looking at all the important factors.
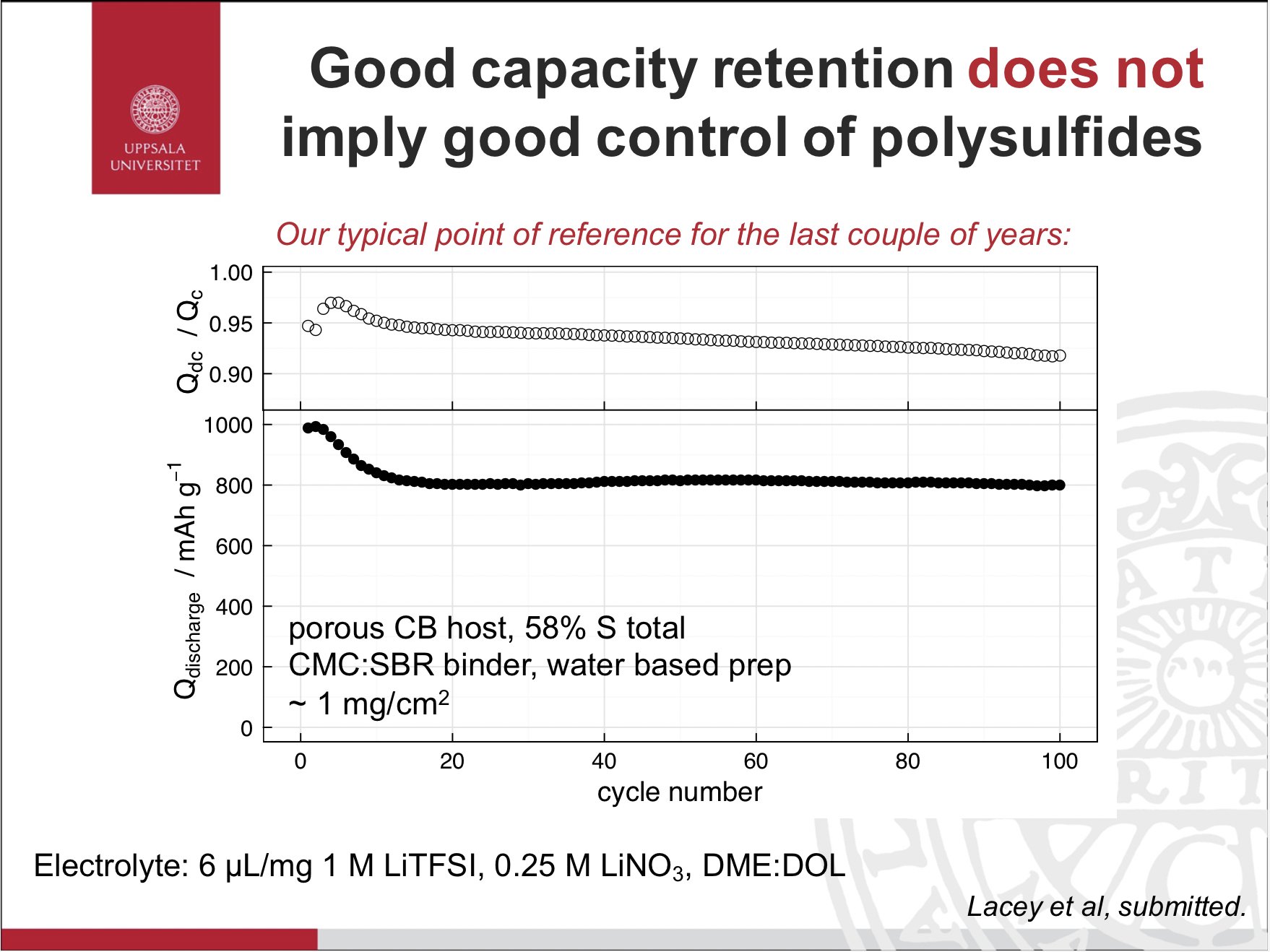
In the Li-S case, you can have dreadful self-discharge from a corrosive catholyte, but it is, to a large extent, reversible. There is an irreversible component too, but as long as you have an excess of lithium… and electrolyte… it’s not as important.
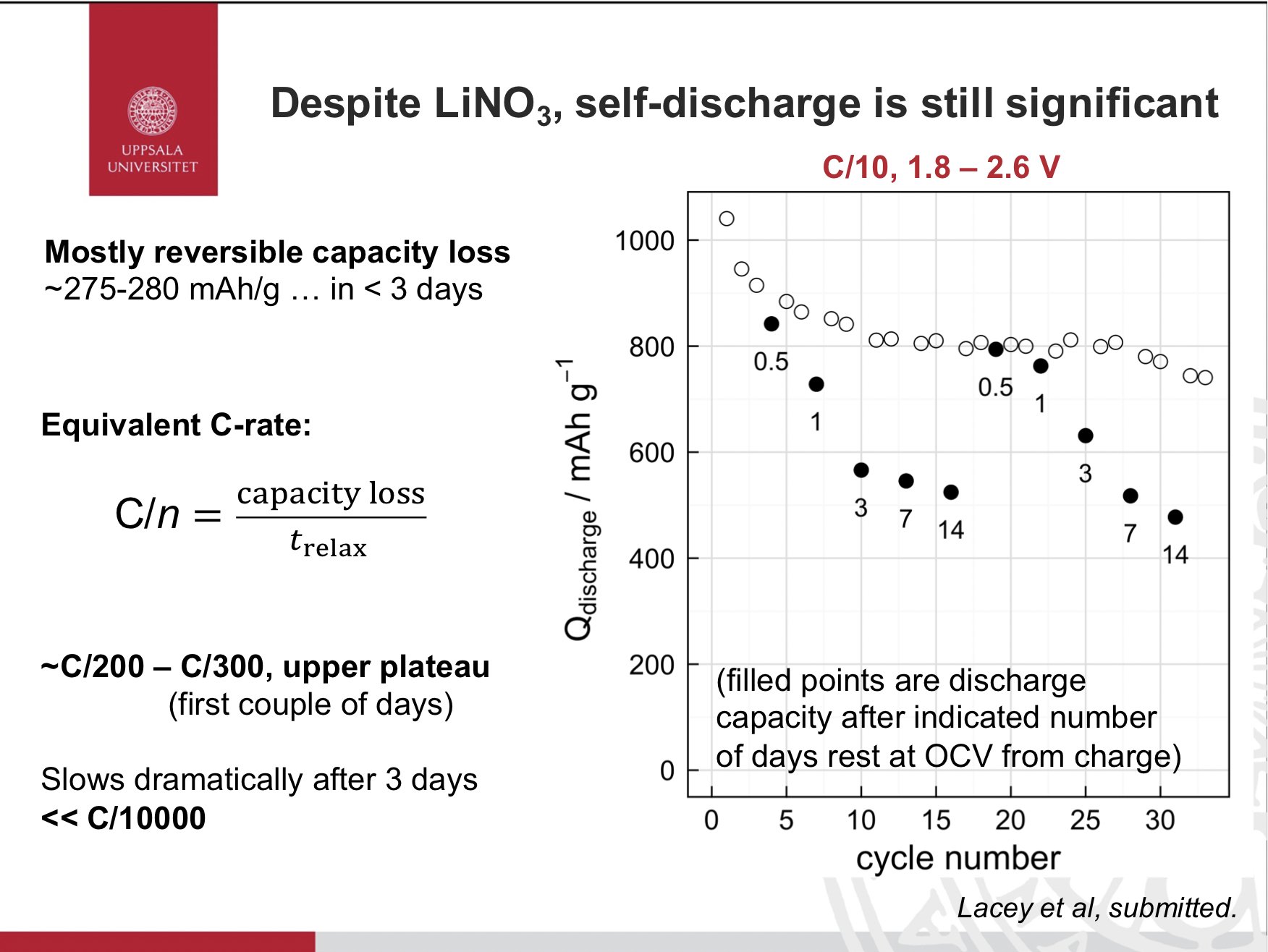
This brings me to my third point. Interfacial stability and its consequences on cycle life, calendar life, self-discharge etc is going to depend on so many factors - materials choices, mutual compatibility, purity, additives, processing, formation steps, cross-talk…
Li-S is one extreme here. Aggressive electrolyte, but generally with an excess of Li metal, which provides a buffer of sorts against Li metal corrosion. Anode-free solid state, such as that of QuantumScape, is at a different extreme.
In that case, 2% of the Li reacting with a purely solid electrolyte would be directly 2% capacity loss and likely produce a highly resistive interface - in other words a disaster.
If that was the case, it would surely have been noticed long ago, so I can fully believe they and those working on other solid state systems, don’t see such drastic Li corrosion. Liquid electrolytes, esp. with Li metal excess, I think sits somewhere between these extremes.
So, to try and sum up… we’re not fighting… I think :) The Nature Energy paper made the very interesting point that good average efficiency is not well-correlated with low electrode self-discharge/good cycle life and this is something to pay attention to.
That said, I think better words could have been chosen to put in the abstract than “regardless of the electrolyte chemistry”. It is always more complicated…
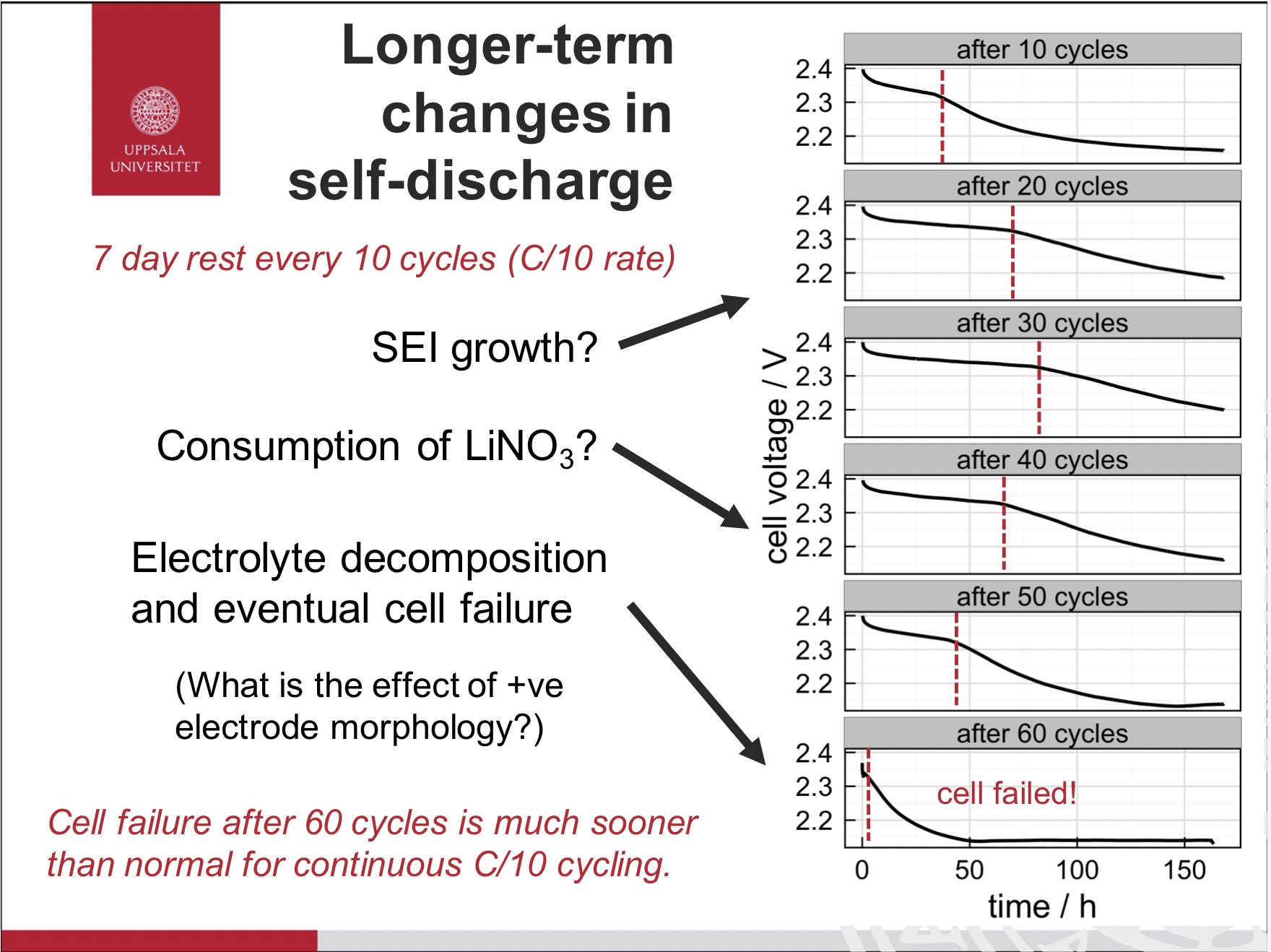
PS. related result I presented in 2015… and never published - self-discharge slowing down and speeding up over time. Also shortened cycle life with a lot of idle time… probably that irreversible component to the self-discharge catching up in the end!