I’m a bit late to this now, but amongst last week’s flurry of solid state battery news was Solid Power announcing a new cell and releasing some cycling data, and of course there were a lot of comparisons to be drawn with QuantumScape. So let’s dig in…
Some of my thoughts already made it into this new article from Steve LeVine, which is well worth a read, but I want to go more into my reasoning…
First, SP’s announcement that they can now produce 20 Ah solid-state cells at 330 Wh/kg with roll-to-roll processes is very impressive. Where QS have so far only shown data from much smaller single-layer cells, SP are showing “the real” thing.
I’m afraid though I struggle to get too excited about the performance, though. Though there’s some promise - preliminary cycling looks quite stable and in a high energy cell this is difficult to manage - other aspects show there’s still plenty of work to be done.
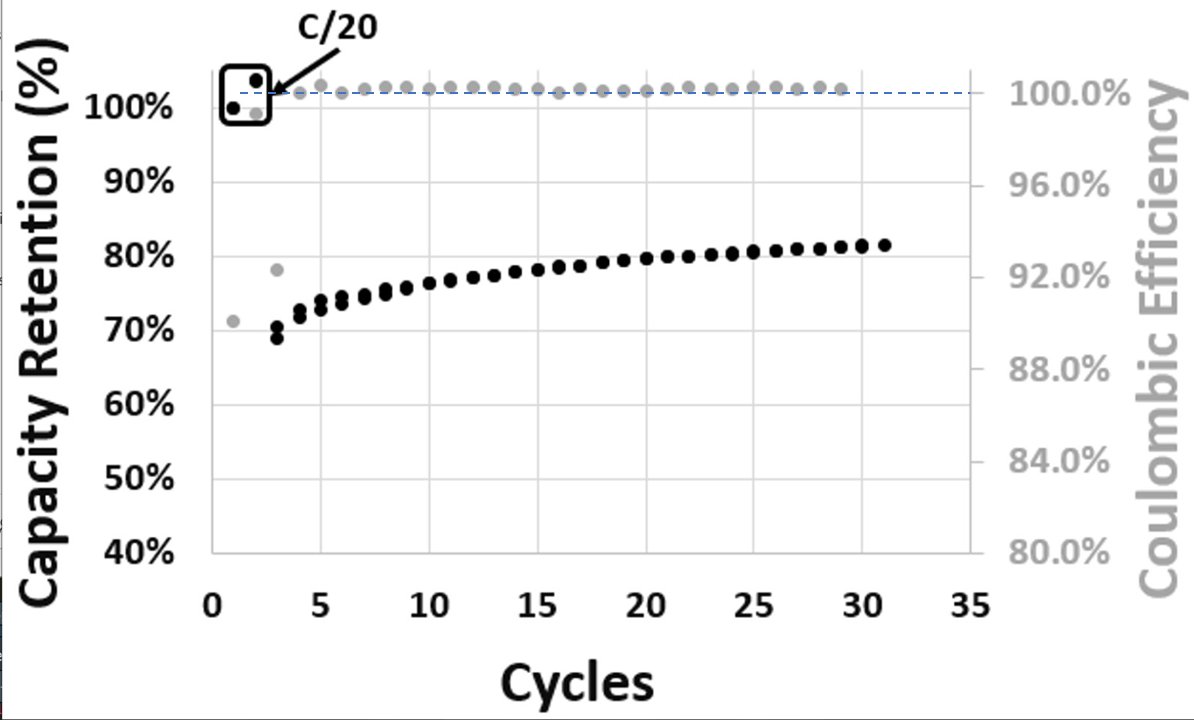
In particular the performance at lower temperatures (<45 °C) is a little disappointing, with very little capacity available at 1C rate.
Continuous cycling at 1C is a typical need for automotive purposes and as others have pointed out, if not addressed it will limit scope of applications.
Obviously, there’s a challenge in reducing cell resistance (which SP fully acknowledge). They have shared small cell data from forthcoming materials showing that this problem is being addressed, but there is still a big gap to be closed to typical Li-ion performance.
A resistance of ~70 Ω cm² on the electrode level is >5x larger than what typical Li-ion provides now (and my eyeball guess of QS data from last week suggests to me they are much closer on this).
I also note how big of a jump this is from the 20 Ah cell data previously - where <10% capacity was accessible at 1C, but with new materials >50% is accessible at 2C - suggests resistance is rather higher in the 20 Ah cell - could also be a problem for thermal management.
It also makes me curious how well SP can currently translate 2 cm² cell performance to the 20 Ah level (i.e., with the same materials and electrode design) - a challenge QS have yet to prove they can do.
I don’t want to speculate much on where the bottleneck might be since I’m not by any expert on the materials. I would guess interfacial resistance (solid-solid contact) and known stability challenges of sulfide electrolytes are in there at least…
One interesting observation on that point. Some hint that average coulombic efficiency is >100%. If it’s not a battery tester issue, might be a sign of an unusual degradation process…
I got asked many times about SP challenges compared to QS, if they can be equated - which I think is difficult. In many ways they have the opposite challenges. QS show glimpses of promising chemistry, but have to scale. SP show scale but chemistry is a challenge.
Doug Campbell was right to point out last week that QS would be wise not to play down the challenge of manufacturing. I’d also point out to those tempted to see the chemistry as the easy bit and manufacturing the hard part to not play down the chemistry challenge.
Sure many startups get stuck with scaling up, but then you’re less likely to see the ones that don’t get past the chemistry phase. There are a few other companies capable of producing similarly sized ~400 Wh/kg cells these days already.
So while I take the point that it’s easy to look at the performance and say it’s not good enough yet, it might be easy to underestimate the challenge of developing the chemistry into something truly competitive. It’s taken Li-ion 40 years to get to where it is now after all!