Wanted to delve into some additional thoughts on the “catholyte” aspect of QuantumScape’s battery tech which has been vigorously discussed in recent days - is it truly solid state, is their data appropriate, what about safety, etc. My take as follows:
First a recap of what the catholyte is - the word means an electrolyte confined to the cathode (positive) side of the cell, and implies a liquid component.
QS have drawn criticism for not calling more attention to this, but it was stated in their presentation and have since confirmed it is a gel, which is also described in some of their patents. (The anode [negative] interface, though, is reportedly purely solid.)
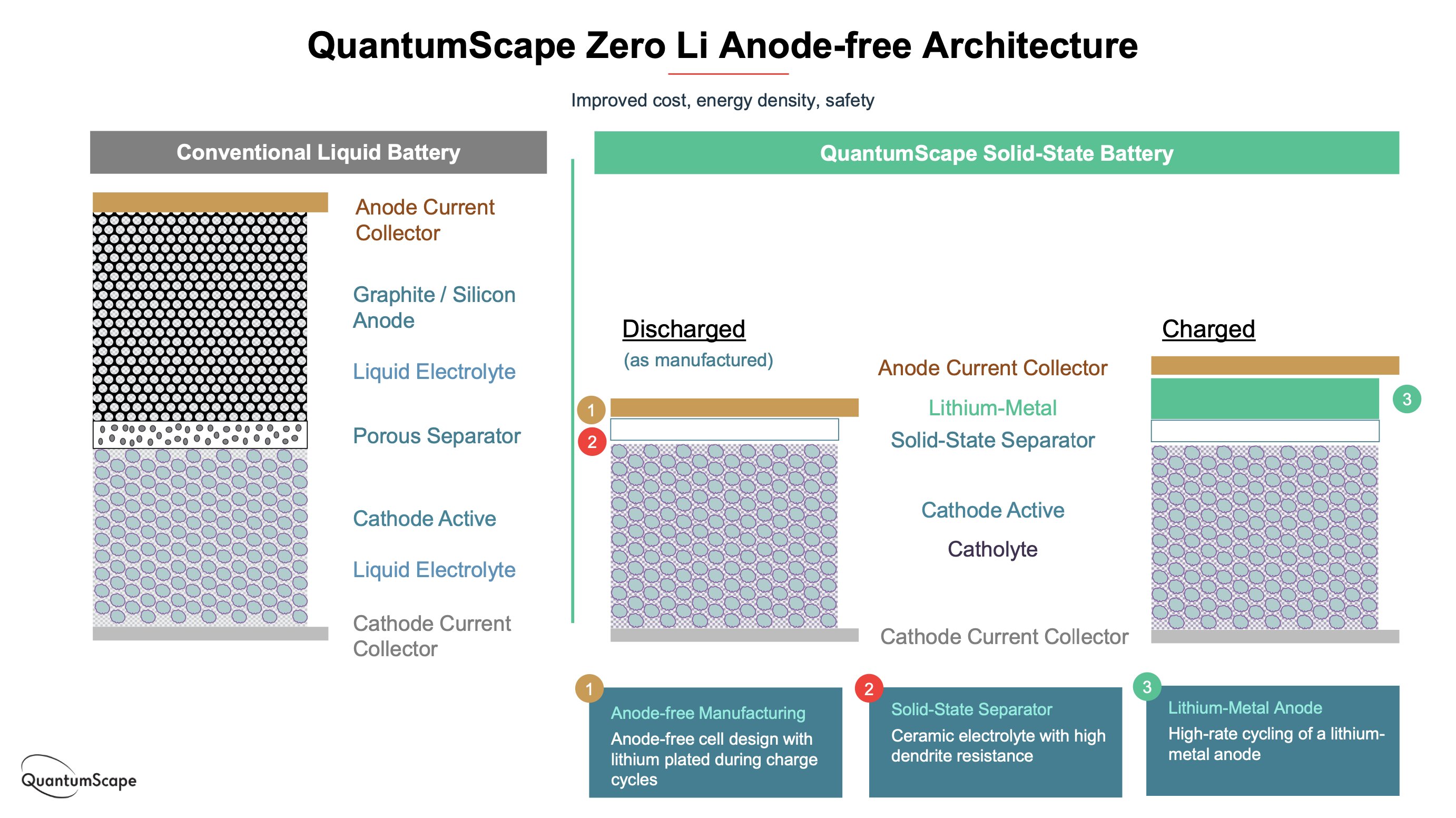
Why use a catholyte? Well, the most obvious reason is that it is much more difficult to get good contact between all the small particles in the porous cathode than it is at the comparatively flat anode.
Without this good contact, the battery would have a much higher internal resistance, which would strongly affect especially power, which is crucial for e.g. automotive applications.
There’s an obvious question then - is QS really making solid state batteries?
It’s obviously not an “all solid state” battery if there’s a liquid component. But solid state has become associated with certain performance gains, esp. energy density and safety, so if it’s not fully solid state, are QS misleading about their technology’s benefits?
The safety question is perhaps the most important. Many have pointed out that the catholyte is still a flammable liquid that can burn in a thermal runaway, much like in typical Li-ion batteries, which is likely true.
Others were critical of this slide in QS’s presentation, noting that thermal stability measurements should include the cathode/catholyte, to be transparent with data related to safety.
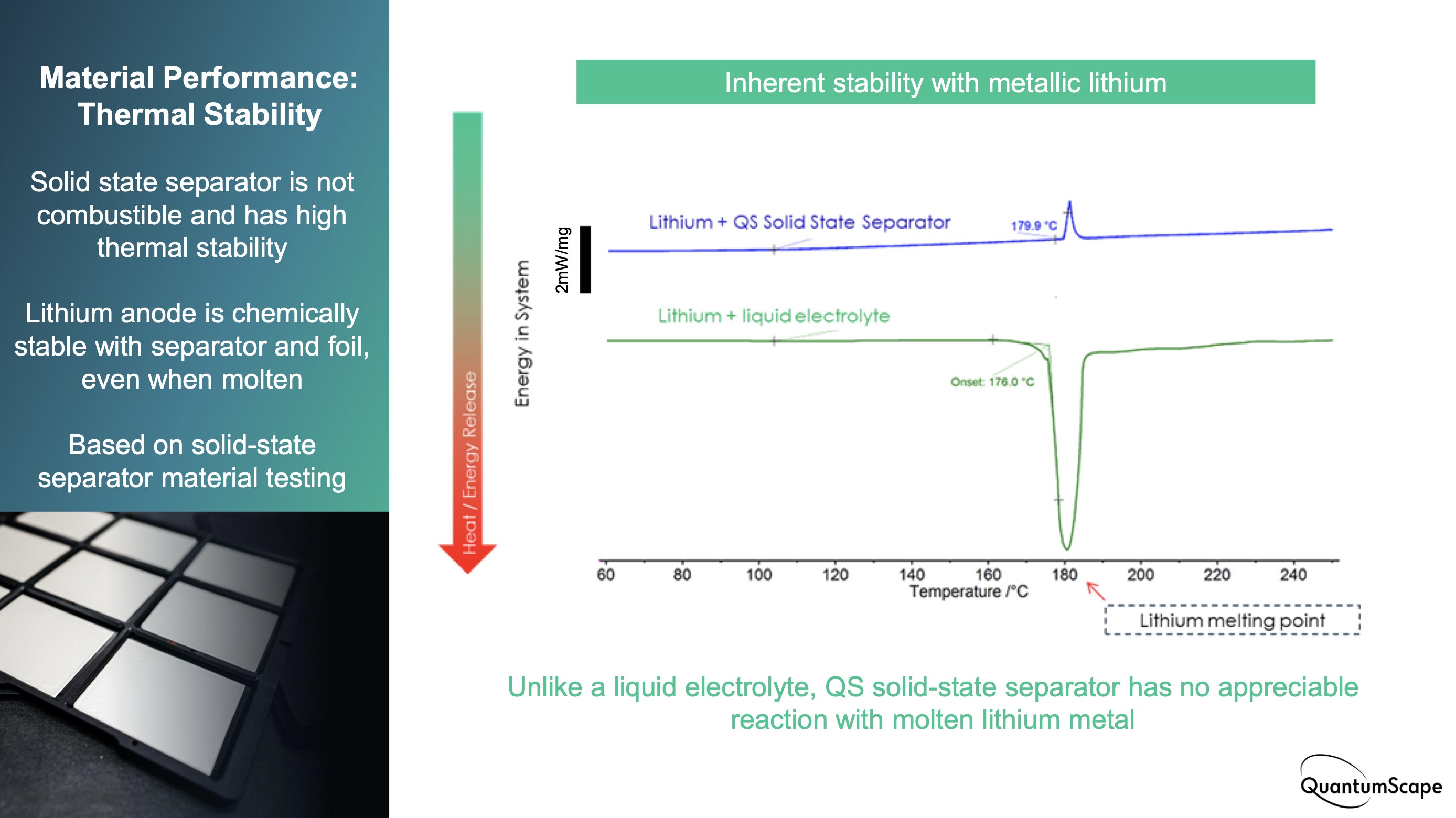
A couple of points on this: first, I didn’t interpret the DSC slide as intending to convey safety-related data (doesn’t mention safety). All I read from it is that the solid electrolyte does not chemically react with Li metal - a good sign from a cycle life perspective.
Though it’s still a good sign the anode/electrolyte interface is stable up to typical thermal runaway temperatures, I think it would be inappropriate to make any safety-related claims anyway, until cells at the intended size are made and can be tested.
Anyway, getting to my point - a generally understood point re. solid state is that you remove the combustible liquids which can burn if the cell is damaged somehow. What’s the point if you still leave some in there? I think the amount of combustible liquid is relevant..
Typical Li-ion cells are getting on for 300 Wh/kg of stored electrical energy now. But about 15% by mass is electrolyte, which if burned releases something approaching 10,000 Wh/kg of energy (estimate - I don’t have exact figs to hand).
So there’s up to ~5 times the potential energy from burning electrolyte as there is stored in the active materials. Not all of this is released in a thermal runaway, but typically the majority of the energy in a TR is burning electrolyte.
QuantumScape’s approach keeps some of this in the catholyte. But it eliminates it in the separator, the anode side, and probably the excess that most Li-ion batteries typically require. So the amount in the cell is reduced drastically - maybe up to 75% or more.
This is maybe not enough on its own to stop a TR and make a cell “intrinsically safe”, but for automotive purposes it may be enough to make it far easier to prevent propagation to neighbouring cells in a pack - making the overall system safe, still a big advantage.
Obviously an all-solid system would likely be even better, but presumably (at least for now) QS consider this a compromise worth making.
IMO, whether or not this ‘counts’ as SS or if it’s even a new concept is academic, what matters is if in the end it delivers a better battery.
As I wrote the other day, chemistry-wise the concept looks very promising to me and I’m keen to see how it develops.
Manufacturing seems to be the big challenge, and QS are playing down the difficulty of this - time will tell of course.
I know some people will consider QS to be misleading on this anyway - that’s fine. I don’t think they’re sugarcoating any more than a typical startup, in an area where it’s been easy to expect too much and get carried away for quite some time. But that’s just my opinion…